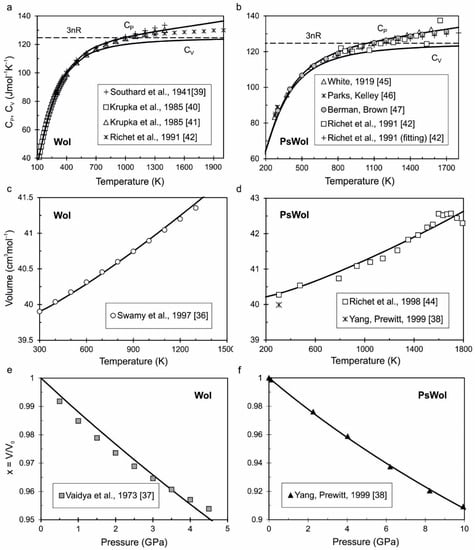
Minerals | Free Full-Text | Equations of State of Ca-Silicates and Phase Diagram of the CaSiO3 System under Upper Mantle Conditions

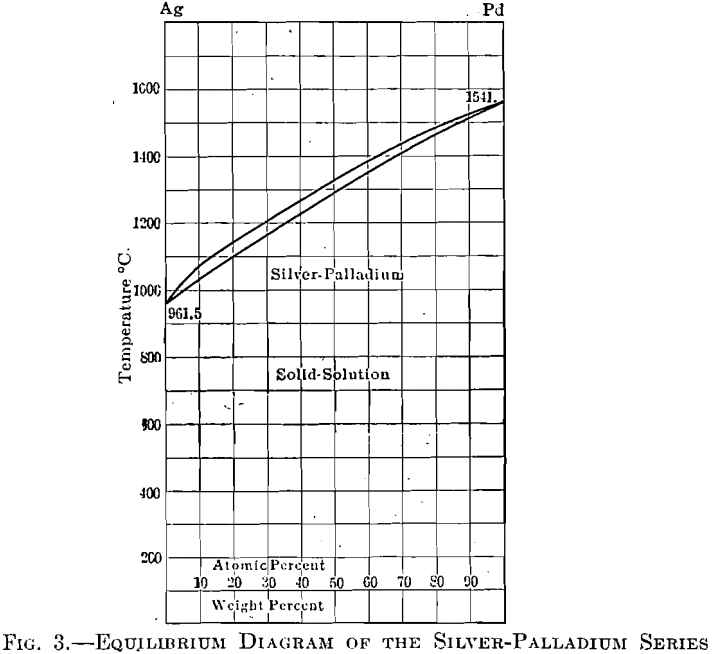
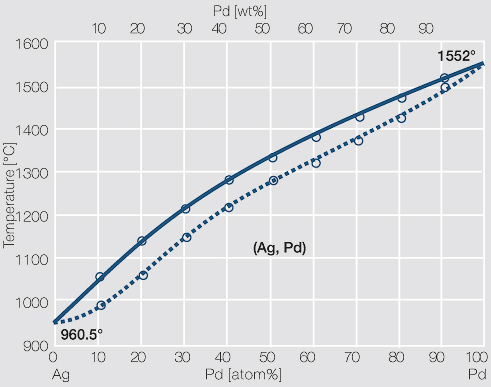
The calculated Ag-Pd phase diagram with experimental points (Ref 4, 5) | Download Scientific Diagram
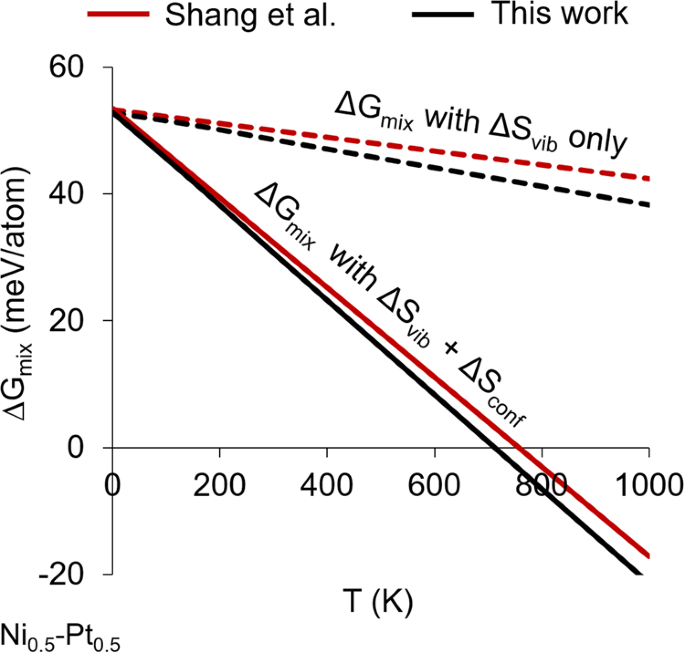
Entropy contributions to phase stability in binary random solid solutions | npj Computational Materials
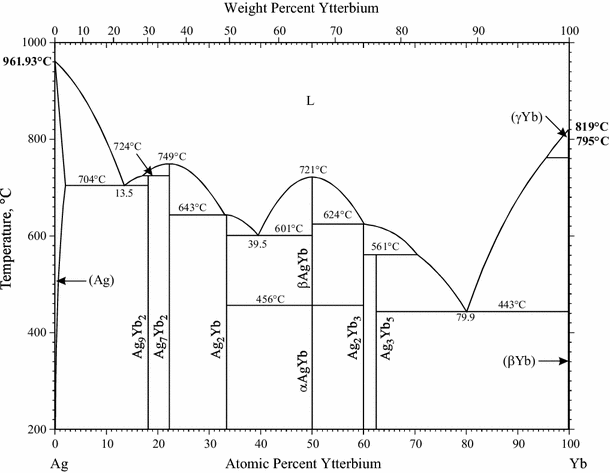
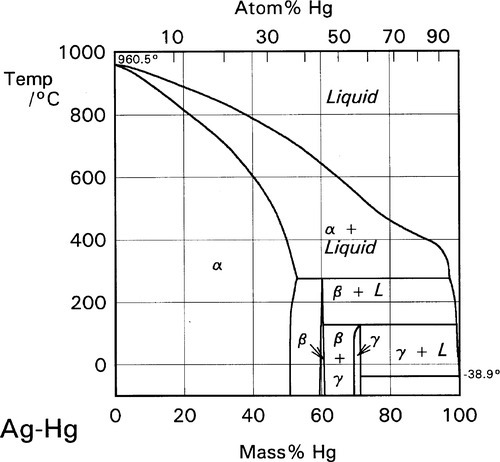
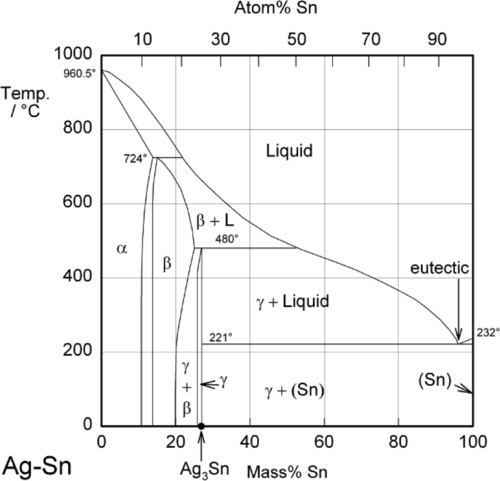
Supplemental Literature Review of Binary Phase Diagrams: Ag-Yb, Al-Co, Al-I, Co-Cr, Cs-Te, In-Sr, Mg-Tl, Mn-Pd, Mo-O, Mo-Re, Ni-Os, and V-Zr | SpringerLink
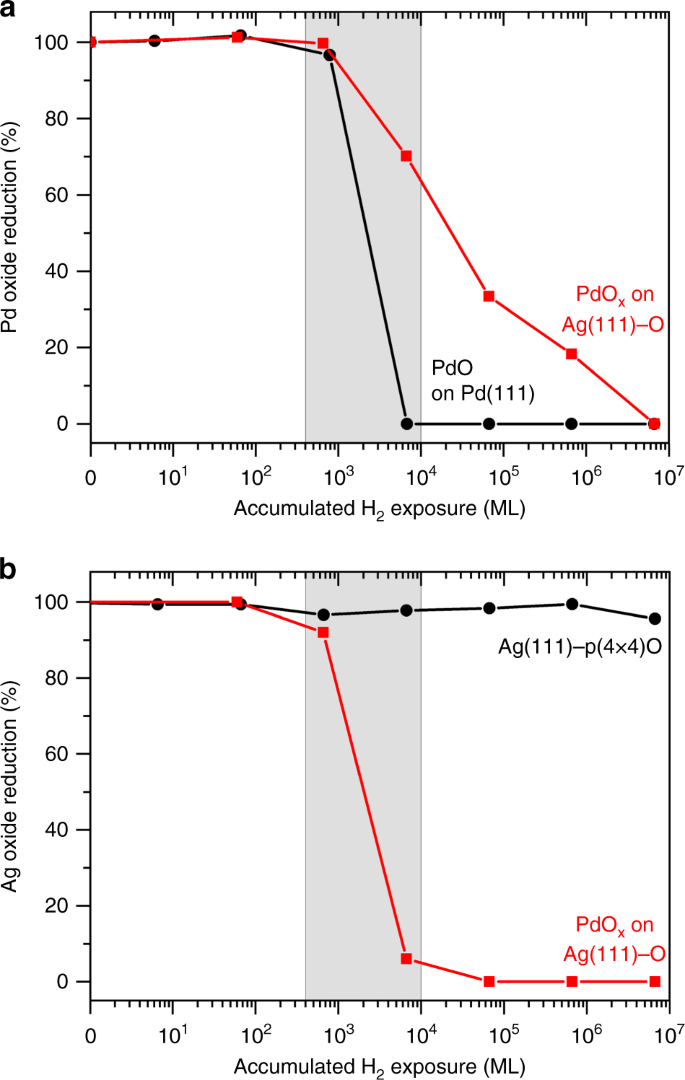
Hydrogen migration at restructuring palladium–silver oxide boundaries dramatically enhances reduction rate of silver oxide | Nature Communications

Supplemental Literature Review of Binary Phase Diagrams: Ag-Co, Ag-Er, Ag-Pd, B-Ce, Bi-La, Bi-Mn, Cu-Ge, Cu-Tm, Er-Y, Gd-Tl, H-La, and Hg-Te | SpringerLink
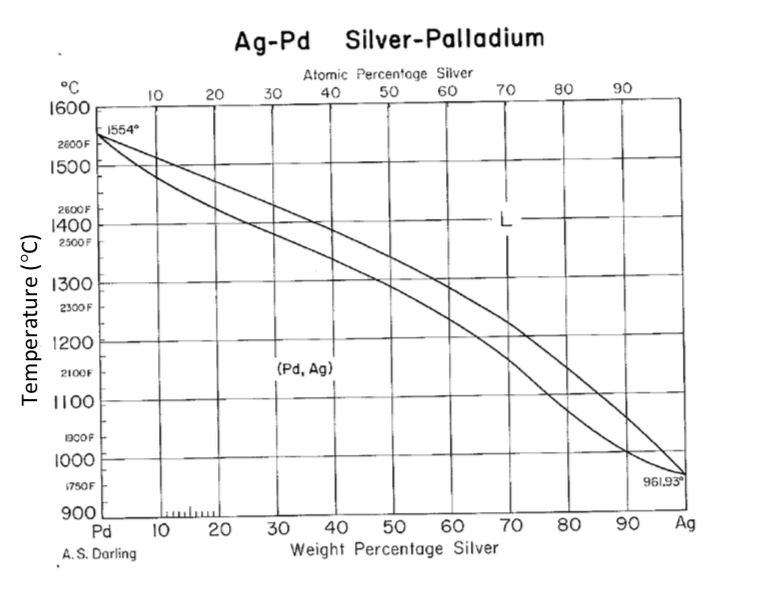
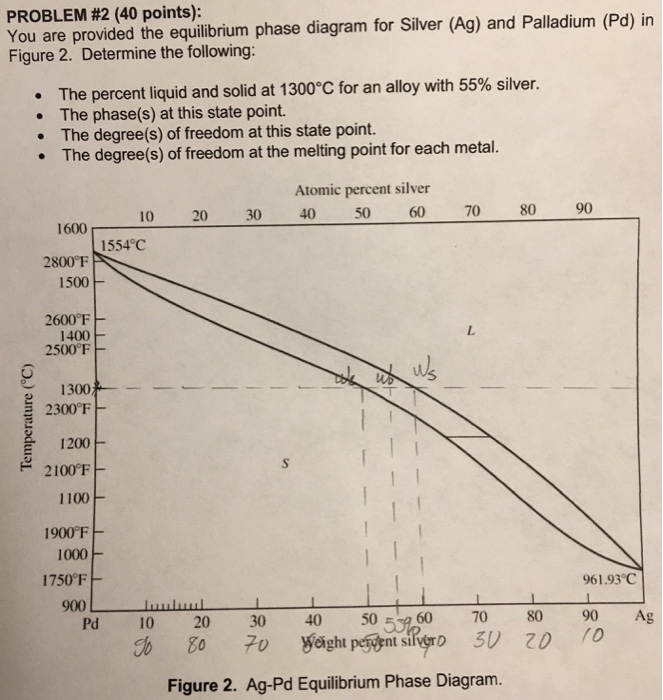
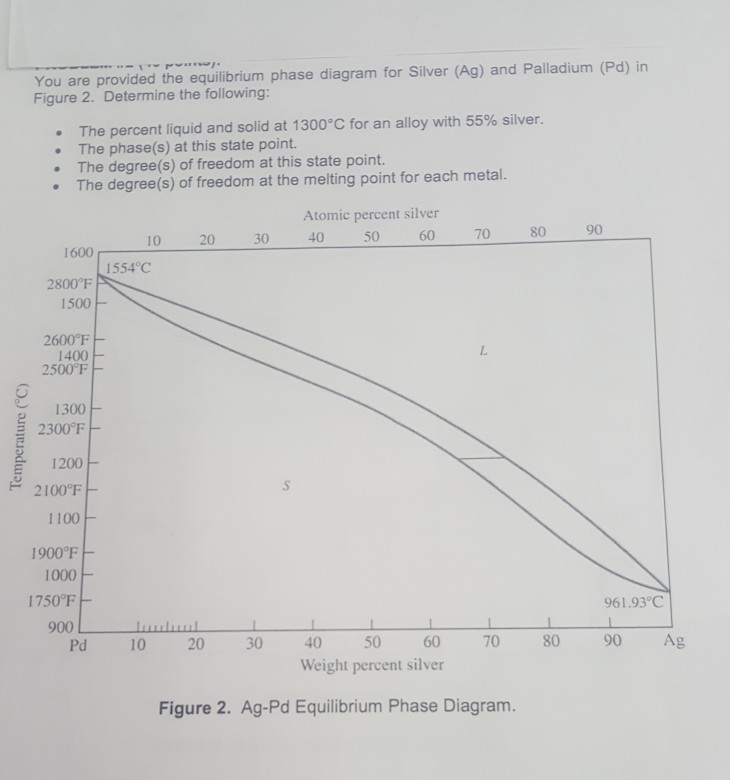
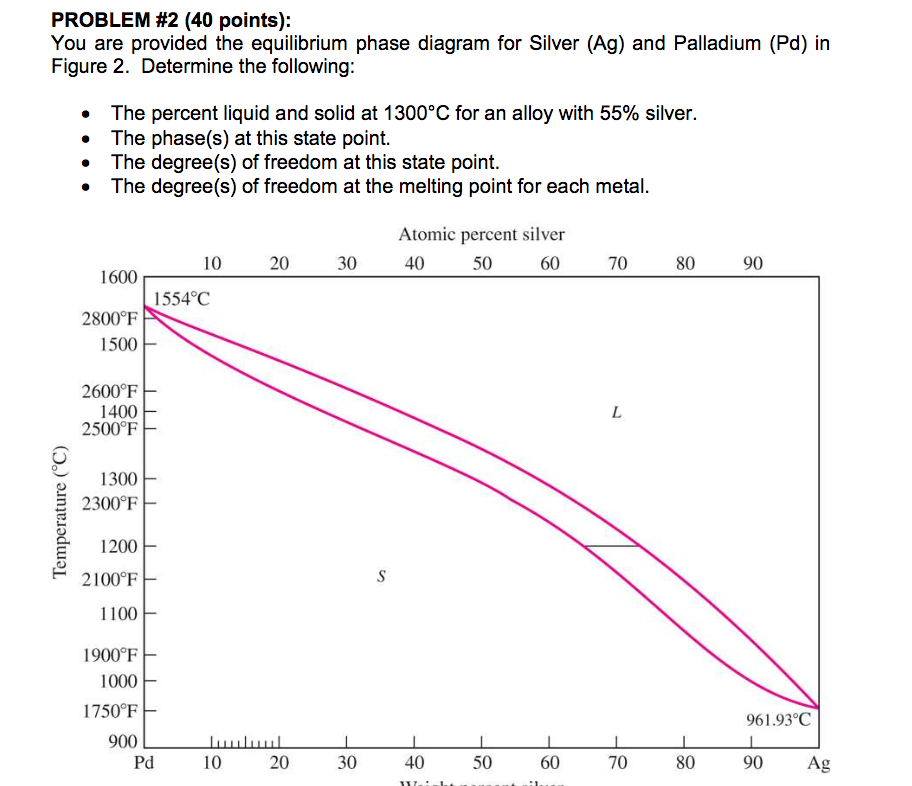
4: The phase diagram for Ag and Pd. The "'L"' signifies the aqueous phase. | Download Scientific Diagram

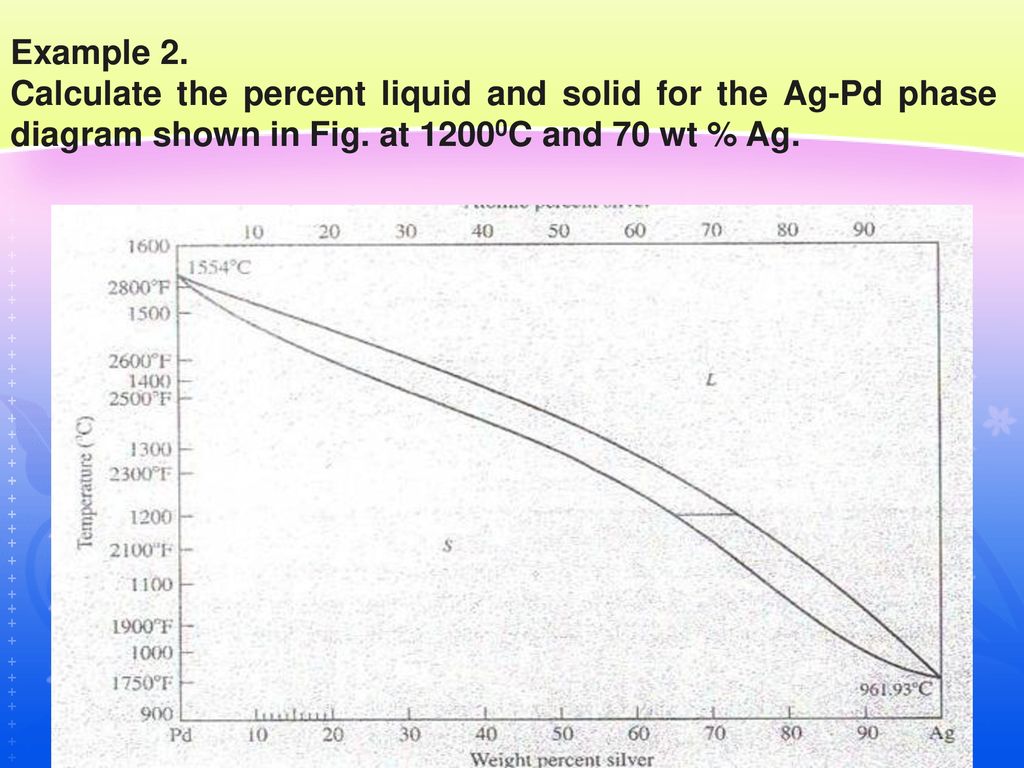
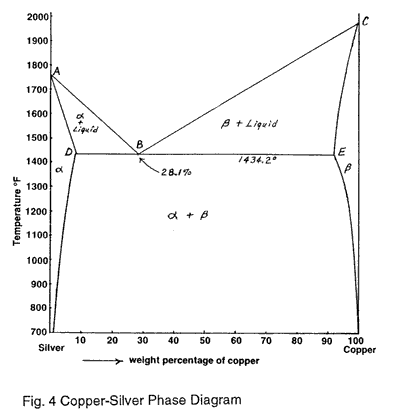
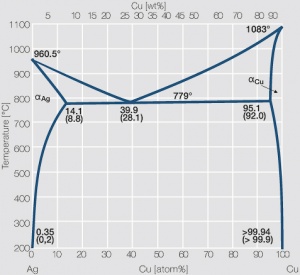
A copper-silver alloy of composition 50 weight % Cu, at equilibrium, is cooled to 600 o C . a) At this temperature, which phases will be present? b) Calculate the approximate mass